A fine-needle aspiration-based protein signature discriminates benign from malignant breast lesions
Bo Franzén, Masood Kamali-Moghaddam, Andrey Alexeyenko, Thomas Hatschek, Susanne Becker, Lotta Wik, Jonas Kierkegaard, Annika Eriksson, Naveen R Muppani, Gert Auer, Ulf Landegren, Rolf Lewensohn
Published in Mol Oncol. 2018
Protein profiling of fine-needle aspirates reveals subtype-associated immune signatures and involvement of chemokines in breast cancer
Bo Franzén, Andrey Alexeyenko, Masood Kamali-Moghaddam, Thomas Hatschek, Lena Kanter, Torbjörn Ramqvist, Jonas Kierkegaard, Giuseppe Masucci, Gert Auer, Ulf Landegren, Rolf Lewensohn
Published in Mol Oncol. 2019
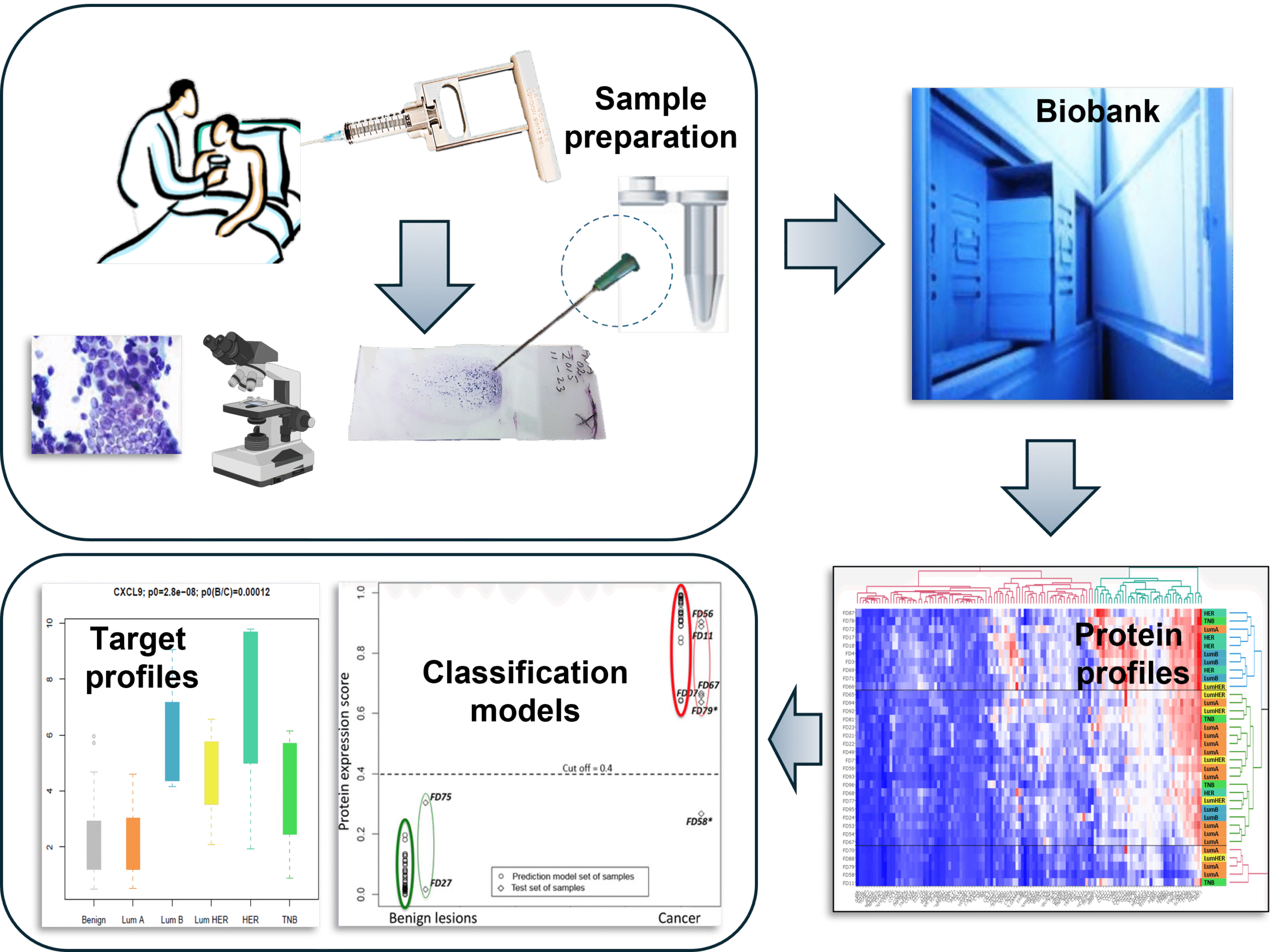
Minimally Invasive Sampling
Fine-needle aspiration (FNA) provides a safe, rapid, and repeatable way to collect tumor material for molecular analysis, even from small or hard-to-reach breast lesions. This approach minimizes patient trauma while enabling real-time diagnostics and longitudinal monitoring.
Multi-Omics Profiling from Tiny Samples
Using proximity extension assay (PEA) and NanoString technology, hundreds of proteins and RNAs, inkluding key RNA markers (ER, PR, HER2, Ki67) can be quantified from minute FNA lysates. These methods deliver high sensitivity and reproducibility, correlating well with routine immunohistochemistry.
Diagnostic and Predictive Signatures
The 2018 study identified an 11-protein signature that perfectly discriminates benign from malignant lesions, while the 2019 study revealed chemokine-rich signatures predicting ER status, Ki67 proliferation, and tumor grade. These tentative signatures outperform traditional binary cutoffs and offer continuous-scale predictions.
Immune Contexture and Tumor Biology
Protein clustering uncovered two major phenotypes: “ER High & Ki67 Low” versus “ER Low & Ki67 High,” with the latter showing elevated chemokines, granzymes, and macrophage infiltration. These findings highlight the role of immune signaling and hypoxia-driven microenvironment changes in aggressive breast cancer.
Clinical Impact and Future Applications
FNA-based multiplex profiling enables rapid, cost-effective diagnostics, supports immunotherapy target identification (e.g., PD-L1), and provides a foundation for AI-driven decision support. This approach paves the way for precision oncology through minimally invasive sampling and comprehensive immune scoring.